Polio Labs
Report by Nellie Bristol and Michaela Simoneau
This fifth installment in our series on U.S. support for global polio eradication discusses the role of the Global Polio Laboratory Network in providing the underlying evidence base for disease detection and identification, and as a model for other networks.
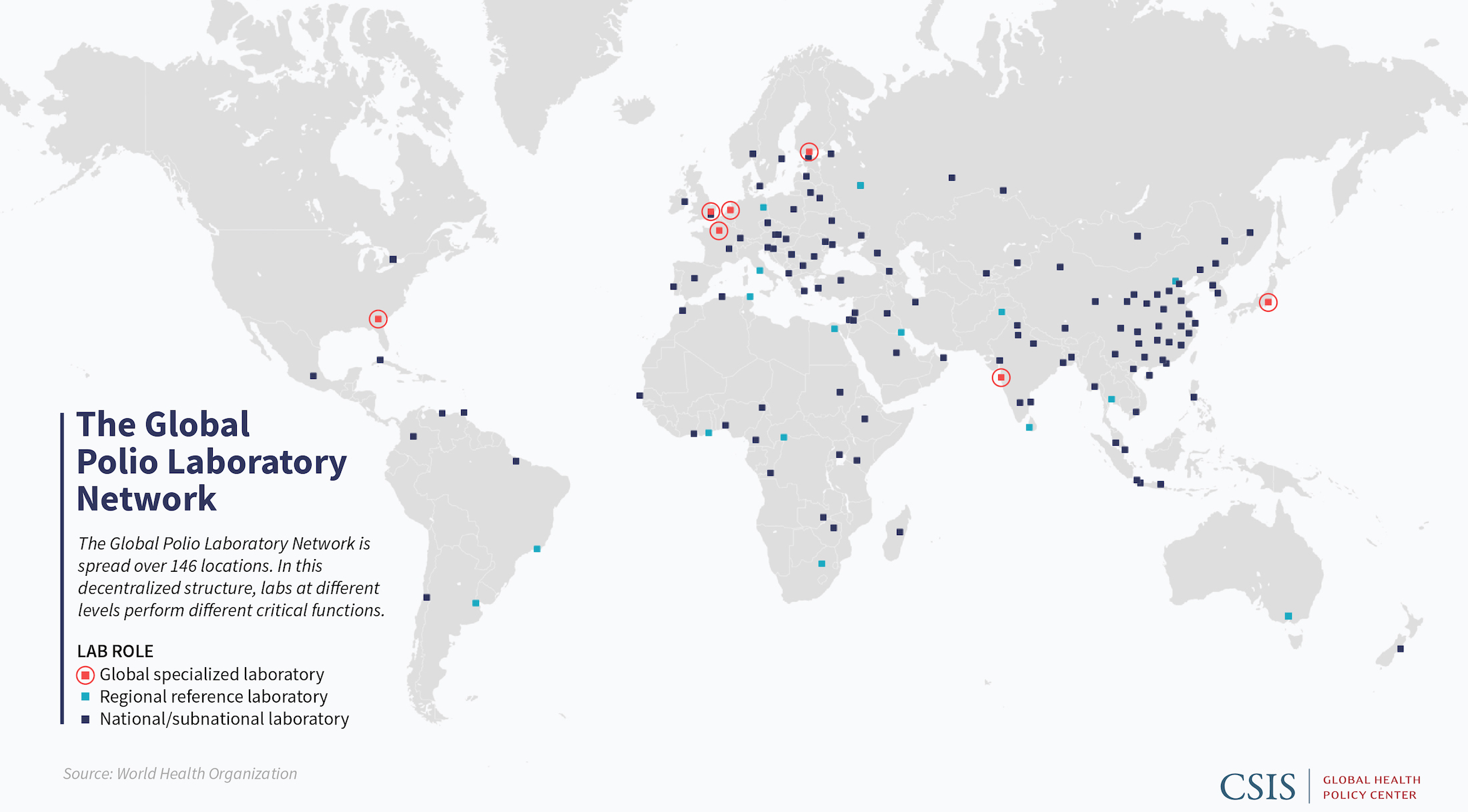
Reliable, standardized data are essential for tracking diseases to their source and understanding their movement among populations. For the Global Polio Eradication Initiative (GPEI), this information is key to judging how the eradication effort is faring and where additional resources and attention are needed. The Global Polio Laboratory Network (GPLN) is critical to making those determinations.
Established in the late 1980s, the now 146-lab network is closely integrated with local surveillance systems, ensuring that every country has access to top quality, near real-time disease identification tools. Public health officials use the data to control outbreaks and plan immunization campaigns. The GPEI, with its focus on finding and eliminating every polio case in the world, has developed the GPLN into a uniquely coordinated and effective system. It is now being used as a model for laboratory networks to help prevent and contain rotavirus, measles, and yellow fever.
Tracking Disease to Its Source
Confirming the presence of disease and identifying its type is the first step to containing it. Through its combined analysis of more than 200,000 stool samples per year, the GPLN determines whether individuals are infected with poliovirus and if so, by which of three serotypes.
It can also determine whether the virus is wild or vaccine-derived. Wild polioviruses are those that occur in nature—only 33 cases of wild virus disease were reported last year in just two countries, Afghanistan and Pakistan. In 2018, there were also 105 reported cases of what is known as circulating vaccine-derived poliovirus. These outbreaks occur when the weakened virus included in the oral polio vaccine passes among a population with low immunity and mutates into a disease-causing state.¹ Knowing which type of poliovirus is causing disease is essential to determining the best response.

A polio sample kit
| WHO Pakistan / S. Mughal
In addition to identifying which type of polio a sample contains, the network conducts genome sequencing analysis to pinpoint where a poliovirus originated. By sequencing the genetic code of a virus, microbiologists can search a database of existing samples to determine what specific strains it is related to. This places the virus’s lineage into a family tree of sorts to help determine its original source.
Comparing this viral genetic information with population movements can show how the virus is traveling from place to place and also suggest containment strategies. For example, through genetic testing, the GPLN was able to trace polio outbreaks in 20 countries in Africa, the Middle East, and Southeast Asia to a 2003 vaccination ban in Northern Nigeria. Armed with this knowledge, leaders and health workers were able to apply diplomatic and epidemiologic resources to halt the ban and contain the virus globally.²

An Effective Collaboration
Efficient operating procedures and a well-trained workforce are critical to the GPLN’s success. In a 2014 survey, GPLN personnel praised the network’s organizational structure, efficient collaboration and coordination among the various levels, integration with immunization programs, competency and reliability of personnel, and excellent quality assurance and data management.³ The value staff place on local ownership and a common culture of high-quality work enables the network to provide data that can quickly be translated into programmatic results in even the harshest conditions.
"There’s a certain institutional culture about these polio labs that’s unique in that when you get in there and you start talking to people, you feel they are all well connected to the program, that they’re all connected to a common goal and that they’re really focused on providing the information in a way the program can use."
–Dr. Paul Rota, Chief, Viral Vaccine Preventable Diseases Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
Like many global polio eradication structures and procedures, the GPLN is based on a model developed for the Americas, the first region to eliminate poliovirus in 1991. Laboratories nominated by national authorities are subjected to on-site evaluations, then judged on their availability of suitable personnel and ability to implement needed procedures. The laboratories are layered into a tiered network that comprises 123 subnational or national members, 16 regional facilities, and seven global specialized laboratories.
Sub-national and national laboratories are the first stop for samples, which have been gathered by polio surveillance officers from potentially infected individuals or found in the environment. Here, the virus is separated from the larger sample and confirmed as poliovirus. Larger regional reference laboratories isolate the virus for countries that do not have their own laboratories, perform viral genetic sequencing, and provide training, quality assurance, and other support for national facilities. The seven global laboratories prepare and distribute reagents needed for testing.

The CDC: Backup Laboratory for the World
While the GPLN is overseen by the World Health Organization (WHO) through regional coordinators, the U.S. Centers for Disease Control and Prevention (CDC) also has played a critical role in its creation and enduring capacity. The CDC serves as one of the seven specialized global laboratories and is the largest with 45 people. The second largest, in India, has 25-30 people on staff.⁴
One of the CDC laboratory’s main tasks is overseeing quality assurance. It manufactures diagnostic and reagent kits for global distribution. These allow all laboratories to conduct the same level of rigorous testing and ensure that results can be understood by public health officials worldwide. CDC also provides human resource support by conducting training workshops and supplying consulting and troubleshooting advice.
"Sometimes if you look at a public health problem and you’re trying to interpret data from a number of different sources, they might be using different techniques and different strategies. It makes it difficult to interpret it. If we have laboratory networks that are functioning under more or less standard operating systems it helps people to interpret what the results mean."
–Dr. Paul Rota, Chief, Viral Vaccine Preventable Diseases Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
While the network’s decentralized structure is focused around regional hubs, CDC plays a role globally as a backup laboratory for much of the world. It performs sample testing for several countries in the Americas and for Yemen, which doesn’t have its own laboratory.⁵ The CDC laboratory also handles genetic sequencing for Nigeria and previously did work for DR Congo.⁶

Monitoring Other Diseases
In addition to analyzing polio samples, GPLN laboratory staff report spending an average of 30 percent of their time providing surveillance data for other diseases, thus aiding national public health systems in identifying and controlling other health threats. There is room for even more expansion. With adequate long-term support the GPLN could play a role in helping to reduce the incidence of diseases ranging from severe acute respiratory syndrome to dengue, to Ebola, to Zika.⁷
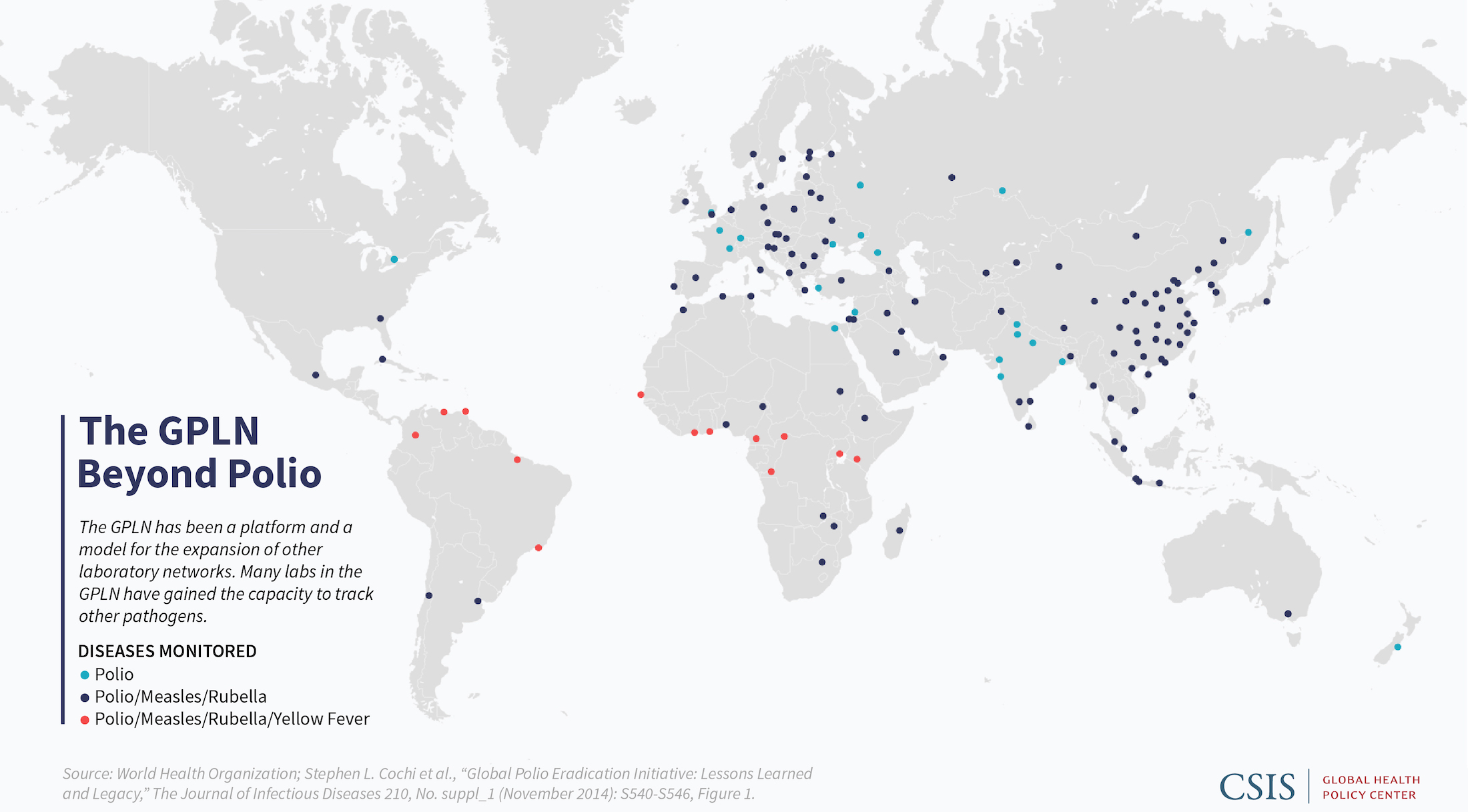
The network is already being used as a model, including for the 700-plus member Global Measles and Rubella Laboratory Network, which uses procedures, training materials, communications strategies, and quality control mechanisms developed by the GPLN for polio eradication.⁸ They also share much of the same infrastructure. As with the polio laboratories, measles and rubella laboratories are designed to create information for action, providing quick analyses to public health officials so they can make timely decisions about their vaccination approaches. Both the GPLN and the measles and rubella network serve as models for a variety of other laboratories including those to monitor yellow fever, Japanese encephalitis, rotavirus, and invasive bacterial diseases.⁹

Sustaining and Integrating the GPLN
"We know how to build these out. It’s just getting the resources to keep them running. It’s challenging. If you look at the measles-rubella lab network, they’re turning out 400,000 test results a year for a couple million dollars. We’re getting a lot of bang for the buck."
–Dr. Paul Rota, Chief, Viral Vaccine Preventable Diseases Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
As with all polio assets, assuring GPLN continuation both for long-term polio needs and to address other health challenges will require political and financial commitment at the national and global levels. A recent study put the network’s cost at $43 million a year. While more than half (62 percent) of the funding for processing samples came from national governments, the remainder was provided by global donors, along with an additional $10 million in external funding to support global and regional coordination.¹⁰ While the goal of the GPEI is for country governments to assume most recurring costs of the polio assets they see as valuable, including the GPLN, many countries will need external financial and technical aid in both the short and the long term.

In fact, the GPEI has identified a set of “polio essential functions”—including long-term polio immunization, surveillance, virus containment, and outbreak preparedness—that outlines what countries will need to maintain for at least 10 years after eradication is achieved. The GPLN will play an essential role in these activities and will require funding from global donors to provide equipment, training, technical aid, and salary support for regional coordinators. In addition, once polio is eradicated, the GPLN will be needed to test environmental samples to ensure a polio-free world continues into the future. It also will be tasked with monitoring a handful of facilities that will retain polioviruses for their work—mainly laboratories and vaccine manufacturers—to ensure the virus remains properly contained.
"Maintenance of global surveillance of vaccine-preventable and other diseases is of the utmost importance for security as well as for health reasons."
–Polio Transition Independent Monitoring Board: A Debt of Honour- Third report, December 2018
Beyond those requirements, with proper planning and support, the GPLN could also serve as the foundation for a surveillance laboratory system for a range of other diseases. The network’s ability to forge interregional relationships is cited as a potential boon to the Global Health Security Agenda, a multi-national consortium aimed at improving global capacities to prevent, detect, and respond to infectious disease.
“There are polio assets that are already being used for other things…you’re just sort of cobbling together from the leftovers, so figuring out how those are going to be supported going forward is going to be critical.”
–Dr. Steve Oberste, Chief, Polio and Picornavirus Laboratory Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
Stakeholders in the laboratory system imagine several options for the GPLN’s continuation: as an organization devoted to specific diseases, integrated into existing laboratory networks, or as the core for broader support of public health activities.¹¹ WHO is leading a process now that will lay out transition strategies for the network and other polio assets.

Preserving and Extending Polio’s Disease Tracking Capabilities
“The relationship between polio laboratories worldwide is quite unique because if you look at every kind of politically complicated region, political issues don’t actually get reflected in laboratory working. Beyond the fact that there are political sensitivities between countries, when we talk about polio eradication, we have the same goal.”
–Dr. Ousmane M. Diop, Coordinator of the Global Polio Laboratory Network, World Health Organization
Through its many donors, including the U.S. government, the GPEI has already made the initial investments in developing a top-notch disease monitoring system with the capacity to not only detect polio but also to contain the spread of other diseases. To maximize that investment, U.S. decisionmakers should proactively support the GPLN to ensure a polio-free world and enhance global health security.
About the Authors
Nellie Bristol
Senior Fellow, Global Health Policy Center

Nellie Bristol is a senior fellow with the CSIS Global Health Policy Center. She leads the Center’s work on efforts to repurpose polio eradication assets for long-term disease control and toward other global health priorities. In addition to an active working group convened to discuss eradication and transition as it relates to U.S. global health policy, she writes extensively on the issue and consults with other organizations focused on transition planning. She also writes about U.S. government relations with multilateral organizations, including the World Health Organization and the World Bank Group. Her major reports for CSIS include Catalyzing Health Gains through Global Polio Eradication, which focused on polio transition in India; Bolstering Public Health Capacities through Global Polio Eradication, which examined polio assets in Ethiopia; The Power of Straight Talk, which looked at the impact of the Independent Monitoring Board on eradication efforts. Bristol came to CSIS following a long career as a health policy journalist. She spent two decades writing about domestic health policy on Capitol Hill before expanding her coverage to global health in 2005. Bristol has written for top publications in the field including The Lancet, Health Affairs, and Congressional Quarterly, covering HIV/AIDS policy, foreign aid and national security, noncommunicable diseases, and efforts to combat maternal mortality. She holds a master’s degree in public health/global health from George Washington University.
Michaela Simoneau
Program Coordinator and Research Assistant, Global Health Policy Center

Michaela Simoneau is a program coordinator and research assistant for the CSIS Global Health Policy Center, where she supports the polio, immunization, and nutrition portfolios. Prior to joining CSIS, she worked as an intern on projects concerning antimicrobial stewardship, conflict resolution, and human rights, and managed her university partnership with a grassroots non-profit organization in Coimbatore, India. Ms. Simoneau holds a B.S. in Biology and International Studies from Boston College, where she wrote her senior thesis on the Rohingya refugee crisis.
Special thanks to:
- Dr. Ousmane M. Diop, Coordinator, Global Polio Laboratory Network, World Health Organization
- Dr. Steve Oberste, Chief, Polio and Picornavirus Laboratory Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
- Dr. Paul Rota, Chief, Viral Vaccine Preventable Diseases Branch, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
- Karen Mason, Associate Director for Policy, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, U.S. Centers for Disease Control and Prevention
This project is made possible through the generous support of the Bill & Melinda Gates Foundation.
A product of the Andreas C. Dracopoulos iDeas Lab, the in-house digital, multimedia, and design agency at the Center for Strategic and International Studies.